Northumbria expands results day support for students
Northumbria University is expanding and enhancing the support it provides to students receiving…
International
Ideally situated in the 5th best student city in the UK (QS Best Student Cities 2026), Northumbria University is a UK Top 40 University (Complete University Guide 2026) with a diverse community of 34,500 students from over 140 countries.
View our Global FootprintBusiness
Northumbria University is proud to offer a range of Professional, Statutory and Regulatory Body (PSRB) approved & accredited courses and programmes. Explore our list of courses and programmes under our Education and Training page.
More on our Business ServicesResearch
Northumbria is a research-rich, business-focused, professional university with a global reputation for academic quality. We conduct ground-breaking research that is responsive to the science & technology, health & well being, economic and social and arts & cultural needs for the communities
Discover more about our ResearchAlumni
Northumbria University is renowned for the calibre of its business-ready graduates. Our alumni network has over 253,000 graduates based in 178 countries worldwide in a range of sectors, our alumni are making a real impact on the world.
Our AlumniThe science of freezing as a preservation technique for food
Pedro Rivero Ramos 09/01/23
Frozen systems within the food industry rely strongly on the glass transition temperature (Tg‘) in order to maintain their quality in fresh-like conditions and expand storage life. In this post, we will learn how this value is key to food scientists and how it allows the identification of the stability of food quality during processing and storage.
Freezing of foods has been in use by humanity for centuries. Initially, however, this phenomenon relied on winter frosts. Farmers, fishermen, and trappers would store and produce in unheated buildings during the winter season. Nowadays, however, the freezing technique itself, just like the frozen food market, is developing to become faster, more efficient and more cost-effective.
Freezing can expand the food shelf life and maintain food stability at subzero temperatures by removing the bulk of water from the solute phase. This phenomenom allows the inhibition of any chemical, physical or biological reaction. However, both the freezing temperature and kinetics are critical to preserving the food quality and texture.
The glass transition temperature (Tg‘) is described as the temperature at which 30-50 carbon chains of a system start to move. At the glass transition temperature, the amorphous regions transit from the rigid state to a more flexible state. Accordingly, at this temperature, the free volume (the gap between the molecular chains) also increases by 150%. It is important to keep in mind that the glass transition phenomenon is a second-order thermodynamic transition. This means that the amorphous material under analysis changes its physical state and not its physical phase. Basically, while a crystalline solid has a long-range molecular order, an amorphous solid lack long-range molecular order but contains short-range molecular order.
Interesting, but how does this occurrence actually translate to the food sector? Fundamentally, because in the glassy state, the motions of molecules within the food matrix are limited to rotations a vibrations only, while significantly reducing the molecular mobility.
During the glassy state, the viscosities of amorphous materials are assumed to show very high values that range between 1012 to 1014 N-s/m2, meaning that the speed of the chemical reactions within frozen foods is strongly decreased, a characteristic that is crucial to ensure successful frozen storage. Contrarily, if the temperature is not low enough to reach values below the glass transition temperature (Tg‘), the molecules within the food matrix will experience translational mobility due to a reduction in the viscosity, generating a liquid-state state. This rubbery state increases the molecular diffusion and the food becomes partially unstable.
Apart from the concept of glass transition temperature (Tg‘), there is also the end point of freezing or onset of melting (Tm‘), an even more crucial value when referring to the stability of foods. This value (Tm‘) is always greater in terms of temperature if compared with Tg‘. If you are a food producer, and your freezers do not cool enough to reach a temperature below the Tm‘of the foods being stored, you are in big trouble, dear reader. At temperatures greater than Tm‘, the molecular mobility is increased significantly to the point where plasticization of melting of ice occurs, resulting in higher rates of diffusion-controlled reactions within the food matrix. The likelihood of the frozen food matrices spoiling would then be high due to poor freezing not reaching lower temperatures.
Therefore, Tg‘ and Tm‘ can be employed to accurately identify the stability of foods at various freezing and frozen storage conditions.
A state diagram is commonly utilised by food scientists for mapping the physical states of food by plotting the temperature as a function of the amount of solids or water content at constant pressure. This diagram would usually display the glass transition curve (which relates to Tg‘ the value), a freezing/melting curve (related to the Tm‘ value) and the condition required to attain maximal-freeze concentration, that is the lowest temperature required to ensure that freezing is achieved when the weigh fraction of solids is relatively high or saturated.
It is noteworthy to mention that both Tg‘ and the Tm‘ values are not the same for all food systems. These values fluctuate strongly depending on the type of amorphous food components present and their molecular weight. Moreover, attention should be paid to the presence and amount of plasticizers. For instance, the higher the molecular weight of food biopolymers (e.g., inulin) within the food, the more reduced the free volume, meaning that both Tg‘and the Tm‘ are increased. However, when the amount of plasticizer increases leads to a reduction in Tg‘ and Tm‘ values. Food researchers, and especially food scientists that perform food quality and safety supervision tasks should be highly aware of the glass transition temperature of the foods with a high-water content that they work with. These values can be accurately used as an indicator to identify stability losses during food production ad storage. Similarly, various characterization techniques can be employed when the glass transitions in frozen systems are not known.
The glass transition temperature in food systems produces variations in the physical properties and behaviour of the materials. These variations are related to the heat capacity, deformation, stability, conductivity, specific volume and electrical resistance. Based on this, food researchers can monitor these changes to further determine the glass transition temperature. Differential scanning calorimetry (DSC), dynamic mechanical analysis (DMA), thermomechanical analysis (TMA), nuclear magnetic resonance (NMR), as well as other emerging technologies are characterisation techniques often reported to determine the glass transition temperature. In future blogposts, I will be explaining how each of these works, but again, they just monitor physical changes in amorphous food materials at different temperatures.
Foods are expected to show the highest stability in the glassy state (achieved by temperatures below Tg‘) due to the low rate of physicochemical deterioration achieved by the diminished molecular mobility. In this blogpost, I quickly reviewed the basics to understand the thermal transition concepts and how pivotal these are for food preservation and stability of foods with high water content. To those who want to learn more about these concepts and the current data on the glass transition temperature of various foods, I strongly recommend reading the review articles displayed in the source materials below.
For more from PhD candidate Pedro please see his blog

Northumbria University is expanding and enhancing the support it provides to students receiving…

With the global population expected to reach 10 billion by 2050 and crop yields declining annually,…

An Assistant Professor at Northumbria University won the Royal Society of Chemistry’s Award…
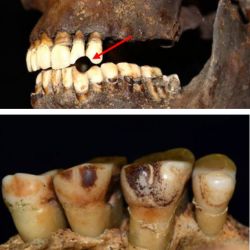
Researchers from Northumbria University have discovered that smokers have tell-tale signs of…

Northumbria University has been named Higher Education Institution of the Year at a prestigious…

Two biomedical sciences researchers from Northumbria University have been awarded grants to…

The Spring 2025 edition of Northumbria University’s newspaper is available to collect on campus…

Scientists have discovered that flies can demonstrate play-like behaviour – the first time…